In the highly regulated pharmaceutical, biotechnology, and medical device industries, maintaining Good Manufacturing Practices (GMP) is not merely a recommendation but a stringent requirement. Regular audits are indispensable tools for ensuring compliance, identifying potential risks, and driving continuous improvement within manufacturing and quality systems. A robust Gmp Audit Report Template serves as the backbone for documenting these critical assessments, providing a structured framework to capture findings, recommend corrective actions, and track their implementation. Without a standardized approach to reporting, the valuable insights gained from an audit can become disorganized, difficult to interpret, and less effective in prompting necessary changes.
The complexity of GMP regulations necessitates a meticulous and systematic approach to every aspect of quality assurance, including the auditing process. An audit report is more than just a summary; it's a legal document, a communication tool, and a roadmap for future compliance efforts. Its effectiveness hinges on its clarity, accuracy, and comprehensiveness. Therefore, having a well-designed template is paramount for any organization committed to maintaining the highest standards of product quality and patient safety.
This article delves into the critical elements, benefits, and best practices associated with using a standardized Gmp Audit Report Template. We will explore how such a template can not only simplify the reporting process but also significantly enhance the overall value and impact of your GMP audit program. From detailing observations to tracking corrective and preventive actions (CAPAs), a good template ensures that every crucial piece of information is recorded consistently and effectively, supporting a culture of quality and continuous improvement.

Understanding the Importance of a GMP Audit Report
A GMP audit report is the formal output of a systematic and independent examination to determine whether quality activities and related results comply with planned arrangements, whether these arrangements are implemented effectively, and whether they are suitable to achieve objectives. In essence, it provides a snapshot of an organization's adherence to regulatory requirements and internal quality standards at a given point in time. The report serves multiple crucial functions, including providing evidence of compliance, identifying areas for improvement, and informing management decisions.

The significance of these reports extends beyond internal reviews. Regulatory bodies often require detailed audit reports as part of their assessment processes, and business partners may request them during due diligence. A poorly structured or incomplete report can lead to misunderstandings, delays, or even regulatory non-compliance, with serious consequences for an organization's reputation and operational license. Conversely, a clear, well-documented report can demonstrate a proactive approach to quality, build trust, and facilitate smooth interactions with stakeholders.

What is a GMP Audit?
A GMP audit is a systematic evaluation of a manufacturing facility, process, or system to ensure compliance with Good Manufacturing Practices regulations. These regulations are established by authorities like the FDA, EMA, and WHO to ensure that products are consistently produced and controlled according to quality standards. GMP audits can be internal (conducted by the organization itself), external (conducted by third-party auditors or customers), or regulatory (conducted by government agencies). Regardless of who conducts it, the objective remains the same: to verify that products are safe, efficacious, and of high quality.

Why Standardized Reporting Matters
Standardized reporting, facilitated by a comprehensive Gmp Audit Report Template, brings numerous advantages. It ensures that all audits are documented consistently, making it easier to compare findings across different audits, track trends over time, and identify systemic issues. Consistency in format and content also simplifies the review process for management and regulatory authorities, as they can quickly locate essential information. Moreover, a standardized template acts as a guide for auditors, ensuring that all critical areas are covered and that observations are recorded in a clear, objective, and actionable manner. Without standardization, audit reports can vary widely in quality and completeness, undermining their utility and potentially leading to overlooked compliance gaps.
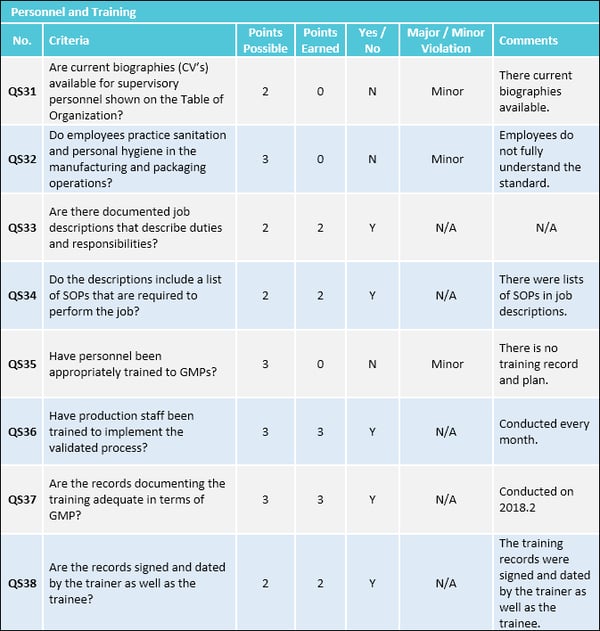
Key Components of an Effective Gmp Audit Report Template
A well-designed Gmp Audit Report Template is structured to capture all relevant information systematically, ensuring that no critical detail is missed. While specific sections may vary slightly based on the industry, scope of the audit, and regulatory requirements, several core components are universally essential. These sections guide the auditor through the reporting process and provide a comprehensive overview for the reader.

Executive Summary and Introduction
This section provides a high-level overview of the audit, including its purpose, scope, and key findings. The executive summary should be concise, highlighting the most significant observations and overall conclusion regarding the auditee's compliance status. The introduction typically reiterates the audit objectives and the specific areas or processes that were examined. This section is crucial for busy executives or regulators who need to grasp the main points quickly.
Audit Scope and Objectives
Clearly defining the audit scope is fundamental. This includes specifying the facility, departments, systems, products, and processes that were subject to the audit. The objectives should state what the audit aimed to achieve, such as verifying compliance with a particular regulation (e.g., 21 CFR Part 211), assessing the effectiveness of a quality management system, or qualifying a supplier. Precise definition prevents scope creep and ensures the report addresses the intended areas.
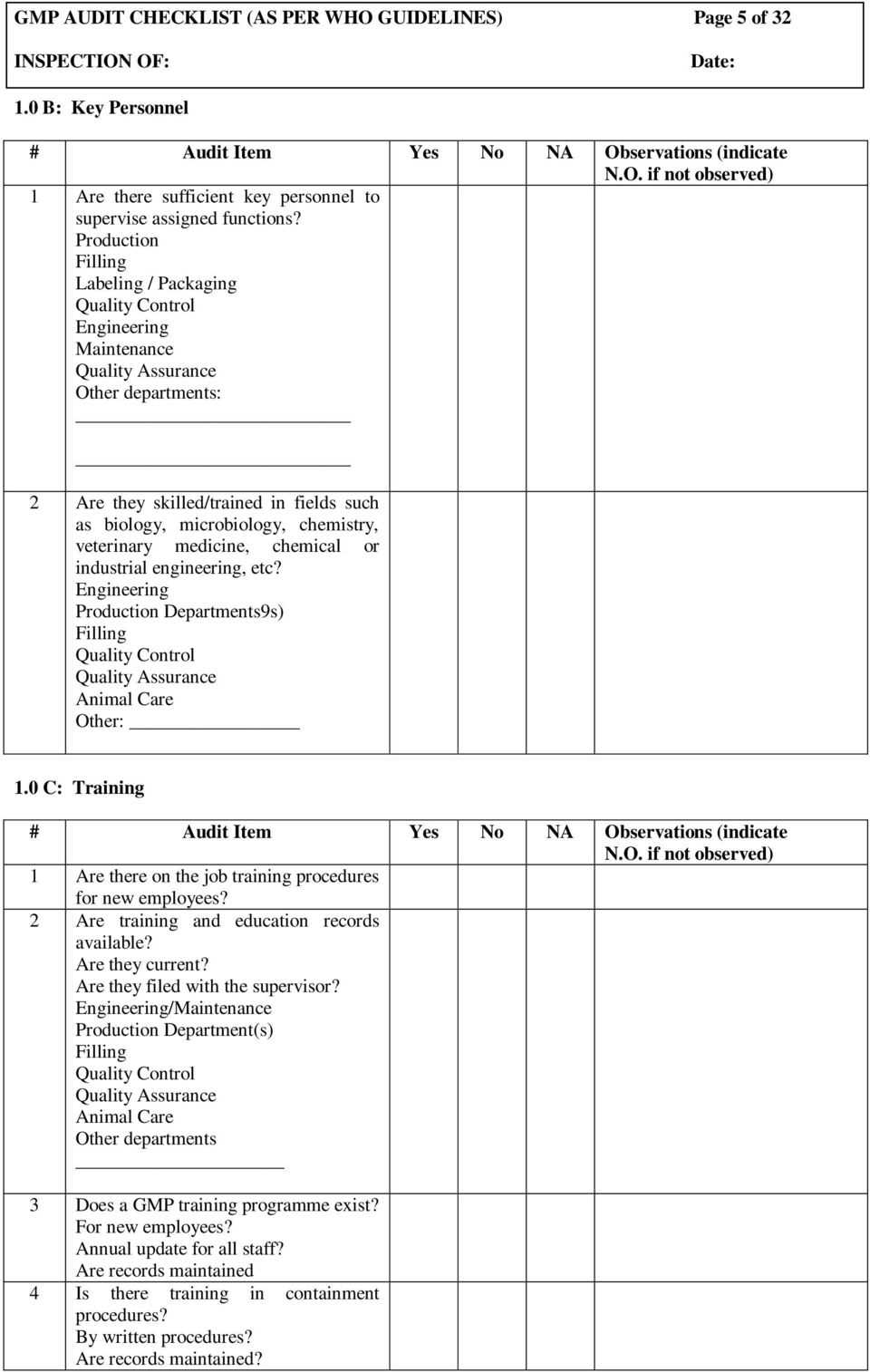
Audit Team and Audited Area
This part of the Gmp Audit Report Template identifies the individuals who conducted the audit, including their roles and affiliations, and the primary contact person(s) at the audited site. It also details the audited area, including the specific location, department, or manufacturing line, and the dates on which the audit was conducted. This provides context and accountability.
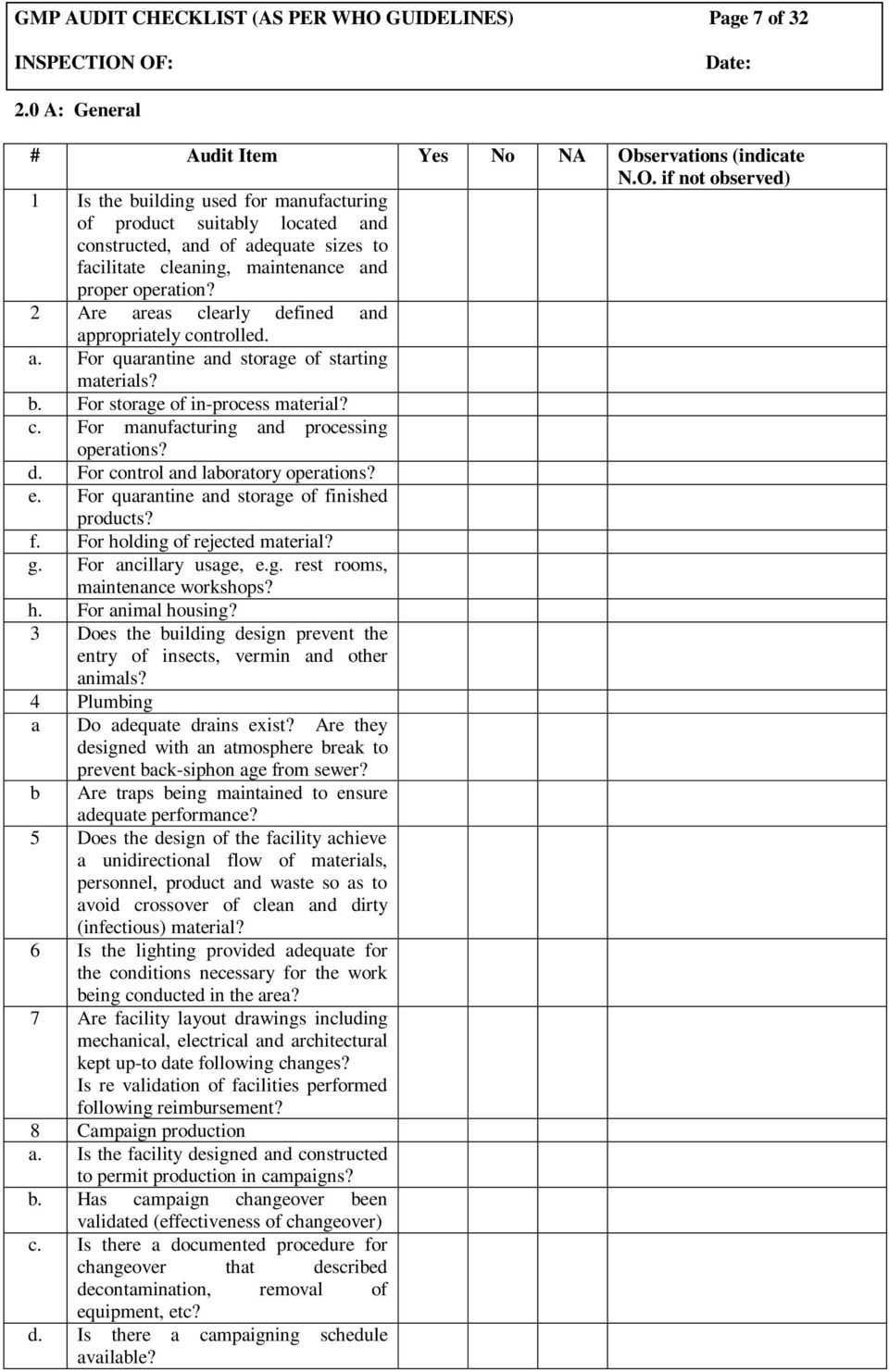
Observations and Findings (Major, Minor, Opportunities for Improvement)
This is often the most substantial section of the report. Here, auditors document their observations and findings in detail, providing objective evidence and references to specific GMP regulations or internal procedures that were found to be non-compliant or effective. Findings are typically categorized to indicate their severity:

- Major Deviations (Critical): Significant non-compliances that pose a direct or serious risk to product quality, safety, or regulatory compliance. These often require immediate action.
- Minor Deviations: Isolated or less serious non-compliances that do not pose an immediate direct risk but indicate a departure from GMP requirements.
- Opportunities for Improvement (OFI): Observations that, while not non-compliances, represent areas where processes or systems could be enhanced to improve efficiency, robustness, or overall quality.
Each finding should be clearly described, stating the specific requirement that was not met and providing factual evidence.

Corrective and Preventive Actions (CAPA)
For each non-compliance identified, the Gmp Audit Report Template should include a section for documenting Corrective and Preventive Actions (CAPA). This section typically outlines:

- Proposed Corrective Action: What needs to be done to address the immediate finding.
- Root Cause Analysis: The underlying reason why the non-compliance occurred (often completed by the auditee after the report is issued).
- Proposed Preventive Action: What steps will be taken to prevent recurrence of the issue.
- Responsible Person: Who is accountable for implementing the CAPA.
- Target Completion Date: The deadline for completing the actions.
- Verification of Effectiveness: How the effectiveness of the CAPA will be confirmed.
While auditors may suggest actions, the auditee is usually responsible for developing and implementing the official CAPA plan, which is then tracked by the auditing party.

Conclusion and Recommendations
The conclusion summarizes the overall status of the audited area's compliance with GMP, based on the findings. It should state whether the site generally complies, requires significant improvements, or is deemed non-compliant. Recommendations provide constructive advice for the auditee to address findings, enhance their quality system, or improve operational efficiency, going beyond just the immediate CAPA requirements.
Appendices and Supporting Documentation
This section houses all supplementary materials that support the audit findings. This can include:

- Copies of documents reviewed (e.g., SOPs, batch records).
- Photographs of observed conditions.
- Interview notes.
- Checklists used during the audit.
- Relevant regulatory guidelines referenced.
- Any other objective evidence collected during the audit.
These appendices provide the detailed evidence necessary to substantiate the claims made in the main body of the report.
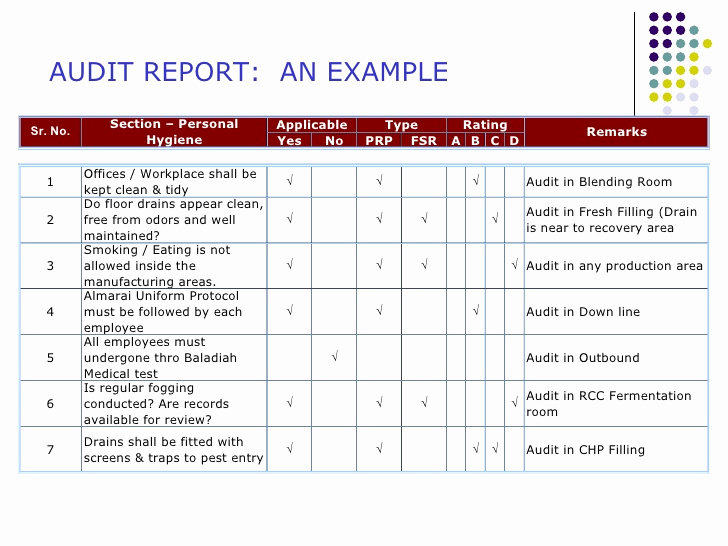
Benefits of Utilizing a Standardized Gmp Audit Report Template
Adopting a standardized Gmp Audit Report Template offers a myriad of benefits that significantly enhance the efficiency and effectiveness of an organization's quality management system. These advantages extend from improved internal processes to better external relationships and regulatory compliance.

Ensuring Consistency and Compliance
A consistent template guarantees that all audit reports across an organization adhere to the same format, scope, and level of detail. This uniformity makes it easier for different auditors to conduct thorough assessments and ensures that critical information is never overlooked. More importantly, it helps maintain consistent compliance with regulatory standards by providing a reliable framework for documenting adherence and identifying deviations from GMP guidelines.

Streamlining the Audit Process
Using a pre-defined template simplifies the reporting phase of the audit. Auditors can spend less time structuring the report and more time focusing on objective observation and accurate documentation of findings. This efficiency reduces the overall time and resources required for each audit, allowing for more frequent audits or better utilization of auditor time. The clear structure also facilitates a smoother review process for management and quality assurance personnel.

Facilitating Root Cause Analysis and CAPA Implementation
The structured nature of a good Gmp Audit Report Template, particularly its dedicated sections for findings and CAPA, naturally guides the auditee towards effective problem-solving. By clearly outlining deviations and their impact, the template supports thorough root cause analysis. This, in turn, leads to the development and implementation of more robust and sustainable Corrective and Preventive Actions, preventing recurrence and driving genuine systemic improvements.
Enhancing Communication and Decision-Making
A clear and comprehensive audit report is an invaluable communication tool. It effectively conveys the audit findings, their implications, and the required actions to all relevant stakeholders, from operational staff to senior management and regulatory bodies. This enhanced communication fosters transparency and enables informed decision-making regarding resource allocation, risk mitigation strategies, and quality system improvements. When everyone understands the issues and the plan to address them, the organization can respond more cohesively and effectively.
Best Practices for Developing and Using Your Gmp Audit Report Template
While a standardized Gmp Audit Report Template offers significant advantages, its effectiveness is maximized when certain best practices are followed during its development and implementation. These practices ensure the template remains a living document that continuously supports and improves the audit process.
Customization and Flexibility
No single template fits all scenarios perfectly. While the core structure should remain consistent, the Gmp Audit Report Template should be flexible enough to be customized for different types of audits (e.g., internal, supplier, pre-approval inspection), different products, or specific regulatory requirements. For instance, an audit of a sterile manufacturing facility might require more detailed sections on environmental monitoring than an audit of a packaging plant. The template should allow for the addition or removal of specific sub-sections as needed, without compromising its overall integrity.
Training and Implementation
It is crucial that all auditors, both internal and external, are thoroughly trained on how to use the Gmp Audit Report Template correctly and consistently. This includes understanding the definitions of different finding categories (major, minor, OFI), the level of detail required for observations, and how to effectively document objective evidence. Consistent application of the template across all audits is key to realizing its full benefits. Clear guidelines and examples should be provided to ensure uniformity in reporting.
Regular Review and Updates
GMP regulations and industry best practices are constantly evolving. Therefore, the Gmp Audit Report Template itself should be subject to regular review and updates. Periodically assess if the template still captures all necessary information, if its structure remains logical, and if it reflects the latest regulatory requirements. Feedback from auditors and auditees can be invaluable in identifying areas for improvement, ensuring the template remains current and highly effective.
Leveraging Digital Solutions
Moving beyond paper-based reports, many organizations are now leveraging digital solutions for their Gmp Audit Report Template. Digital platforms can offer:
- Automated data entry: Reducing manual effort and errors.
- Integrated CAPA management: Linking findings directly to corrective actions and tracking their progress.
- Version control: Ensuring everyone uses the latest template.
- Searchability and analytics: Making it easy to find specific information, identify trends, and generate reports.
- Workflow management: Streamlining the review and approval process for reports.
Digital templates can significantly enhance efficiency, data integrity, and the overall management of the audit process.
Common Challenges and Solutions in Gmp Audit Report Template Usage
Despite the clear advantages, organizations can encounter challenges when developing and utilizing a Gmp Audit Report Template. Anticipating these issues and having strategies to address them can significantly improve the template's effectiveness.
Ensuring Objectivity and Detail
One common challenge is ensuring that auditors maintain objectivity and provide sufficient detail in their observations. Reports can sometimes be subjective, lack concrete evidence, or generalize findings without specific examples.
Solution: Provide rigorous training on how to write objective observations backed by factual evidence (e.g., "SOP 001, Section 3.2, was not followed as observed on Line A, batch record XYZ, page 5, where temperature was recorded as 20°C instead of the required 22-25°C"). Include mandatory fields in the Gmp Audit Report Template for referencing specific documents, dates, and locations. Implement a robust review process for all draft reports by a senior quality professional to ensure clarity, objectivity, and completeness before finalization.
Managing Follow-up Actions Effectively
Another significant challenge is the effective tracking and verification of corrective and preventive actions arising from audit findings. A well-documented report is only valuable if the identified issues are adequately addressed and verified.
Solution: Integrate the Gmp Audit Report Template with a CAPA management system. Whether it's a dedicated software or a well-structured spreadsheet, this system should enable real-time tracking of CAPA status, responsible persons, and due dates. The template should prompt for a mechanism of effectiveness verification for each CAPA. Regular management reviews of CAPA progress and effectiveness should be a standard practice, ensuring that identified issues lead to tangible and lasting improvements. The template can also include a section specifically for auditor follow-up actions and dates, to schedule re-audits or verification checks.
Conclusion
The Gmp Audit Report Template is more than just a form; it is a critical instrument in upholding Good Manufacturing Practices and fostering a robust quality culture within any regulated industry. A well-designed, comprehensive, and consistently applied template is indispensable for documenting audit findings, facilitating root cause analysis, driving effective Corrective and Preventive Actions, and ultimately ensuring product quality and patient safety. By providing a structured framework, it enhances consistency, streamlines the audit process, and improves communication among all stakeholders. Embracing best practices in template development, auditor training, and leveraging digital solutions can further amplify these benefits, transforming audit reports from mere compliance documents into powerful tools for continuous improvement. Organizations that invest in developing and diligently using an effective Gmp Audit Report Template are better positioned to meet regulatory expectations, mitigate risks, and maintain the highest standards of operational excellence.
0 Response to "Gmp Audit Report Template"
Posting Komentar